
Fig. 1. Molecular structure of [Rh(3,6-DBSQ-4-NO2)(CO)2].
Heat capacity measurement of the one-dimensional rhodium(I)-semiquinonato complex [Rh(3,6-DBSQ-4-NO2)(CO)2] was performed by adiabatic calorimetry. A first-order phase transition was observed at 176.3 K. Transition enthalpy and entropy were estimated to be 580 ± 16 J mol−1 and 3.39 ± 0.11 J K−1 mol−1, respectively. The value of this transition entropy suggests that this phase transition is of displacive type.

Fig. 1. Molecular structure of [Rh(3,6-DBSQ-4-NO2)(CO)2].
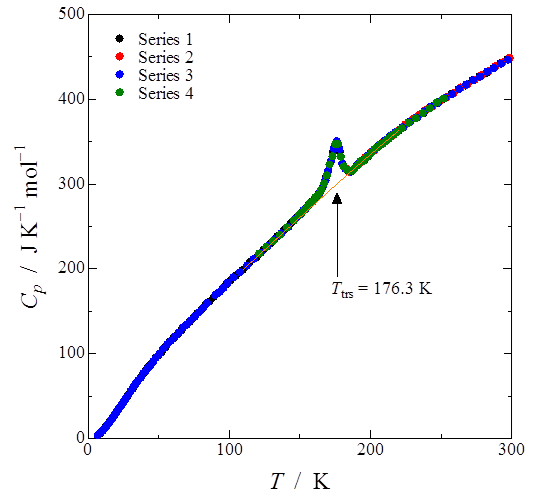
Fig. 2. (Color online) Heat capacity of [Rh(3,6-DBSQ-4-NO2)(CO)2]. First-order phase transition was observed at 176.3 K.
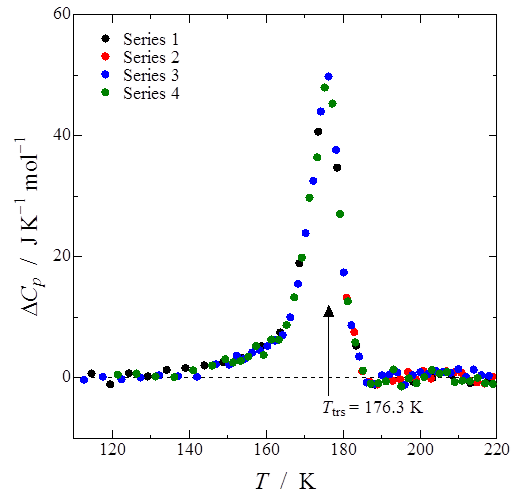
Fig. 3. (Color online) Excess heat capacity of [Rh(3,6-DBSQ-4-NO2)(CO)2]. Transition enthalpy and entropy were estimated to be 580 ± 16 J mol−1 and 3.39 ± 0.11 J K−1 mol−1, respectively.
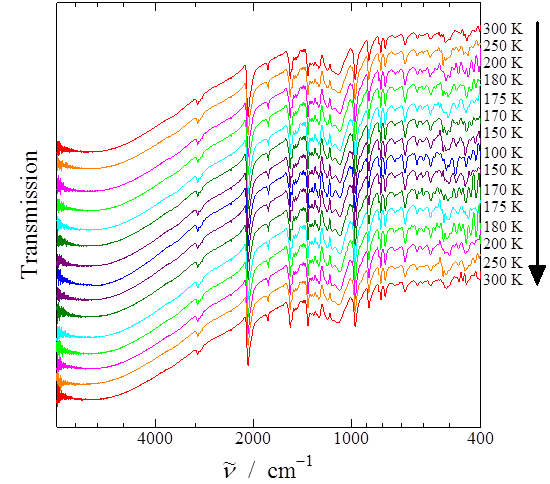
Fig. 4. (Color Online) IR spectra of [Rh(3,6-DBSQ-4-NO2)(CO)2].